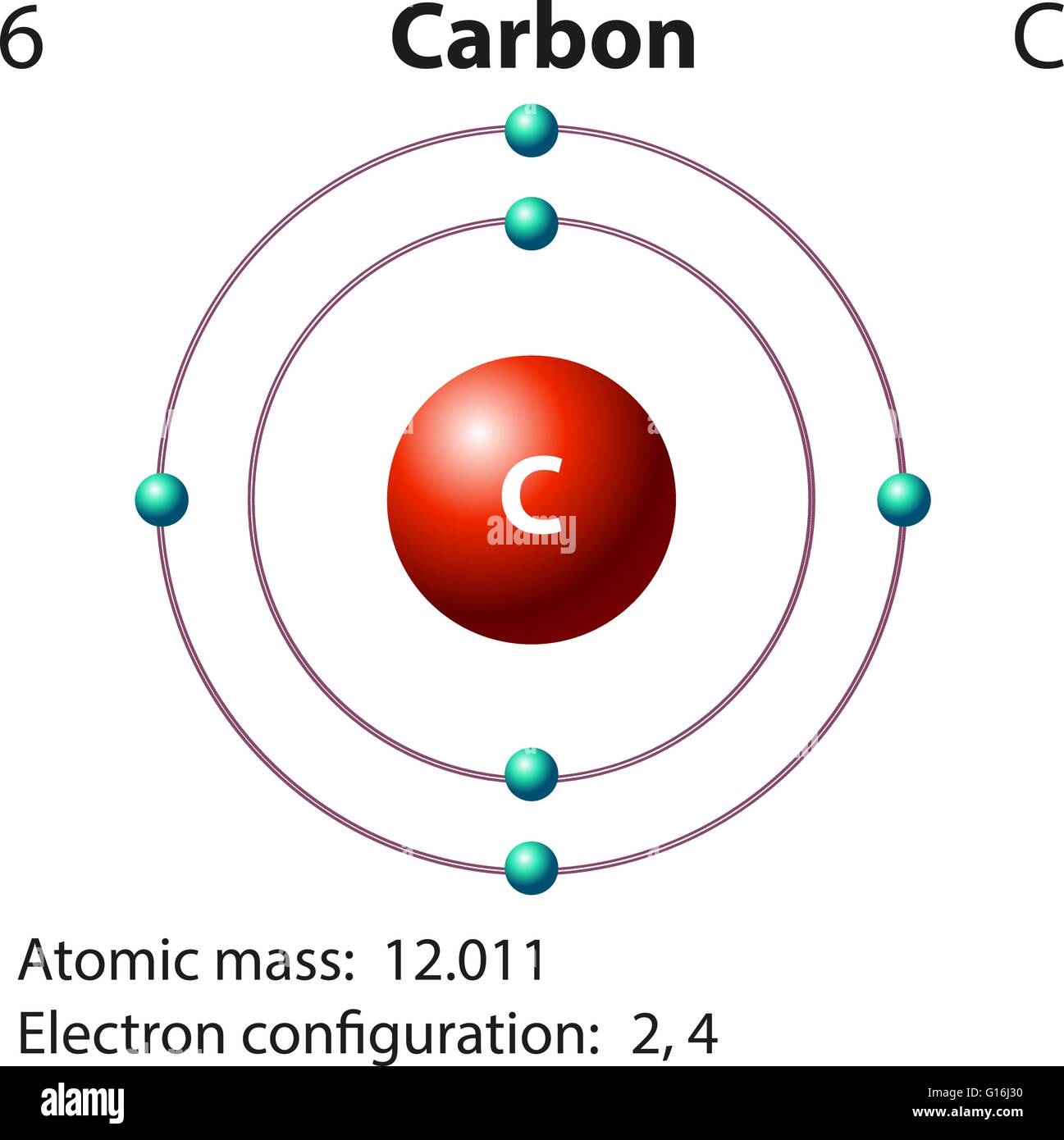
Carbon atom diagram hires stock photography and images Alamy
Electronic Structure of Atoms and Molecules Electronic Configurations

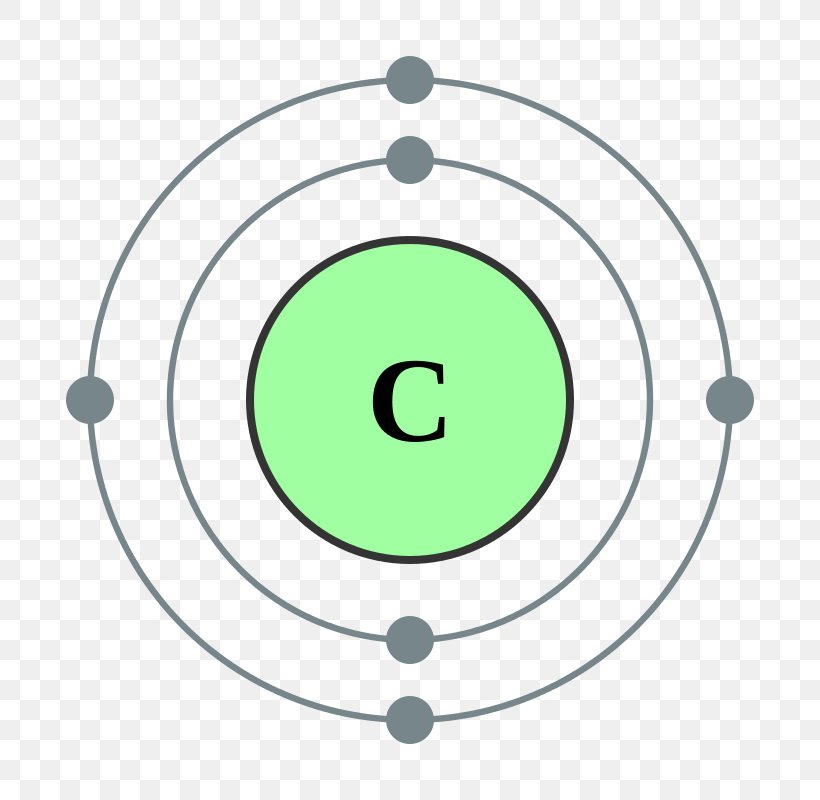
Electron Distribution Diagram Of Carbon
What are electron configurations? The cells in our bodies are masters of quantum physics---they've figured out the complicated dance of atoms and electrons, and they use this knowledge to build an endlessly complex series of signalling pathways and genetic circuits.

Electronic Configuration Explanation & Examples Embibe
An atom's ground state electron configuration describes how the electrons have distributed among the orbital shells and subshells. According to the electron configuration chart , electrons in an atom occupy orbitals according to their increasing energy, with each orbital having a maximum of two paired electrons with opposite spins .
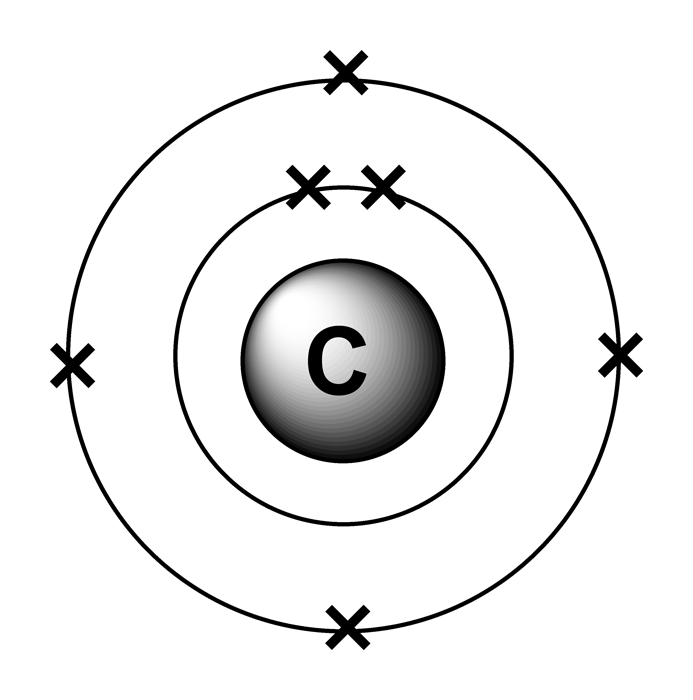
Electron arrangements
Answer: The electron configurations of the elements are presented in Figure 2.2.3, which lists the orbitals in the order in which they are filled. In several cases, the ground state electron configurations are different from those predicted by Figure 2.2.1. Some of these anomalies occur as the 3 d orbitals are filled.
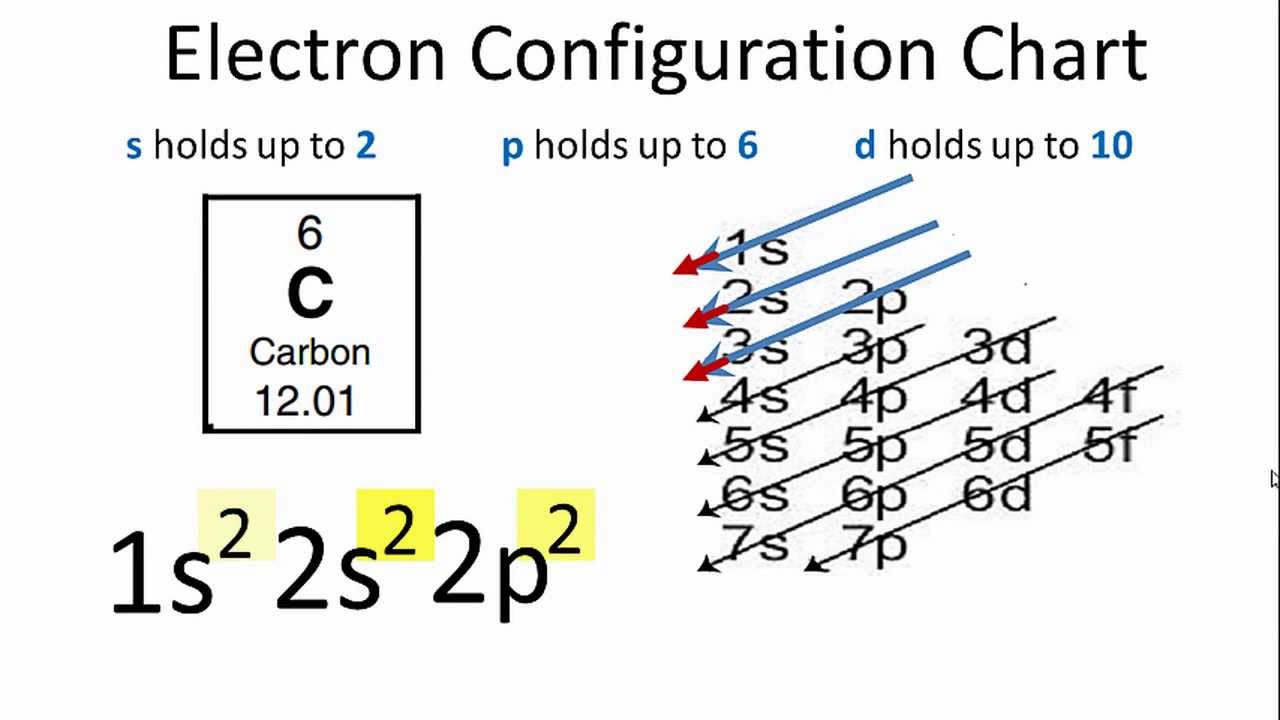
Carbon Electron Configuration YouTube
Electron configurations The ground-state electronic configurations of atoms of these carbon group elements show that each has four electrons in its outermost shells. As has been explained, if n represents the outermost shell ( n being two for carbon, three for silicon, etc.), then these four electrons are represented by the symbols ns2np2.
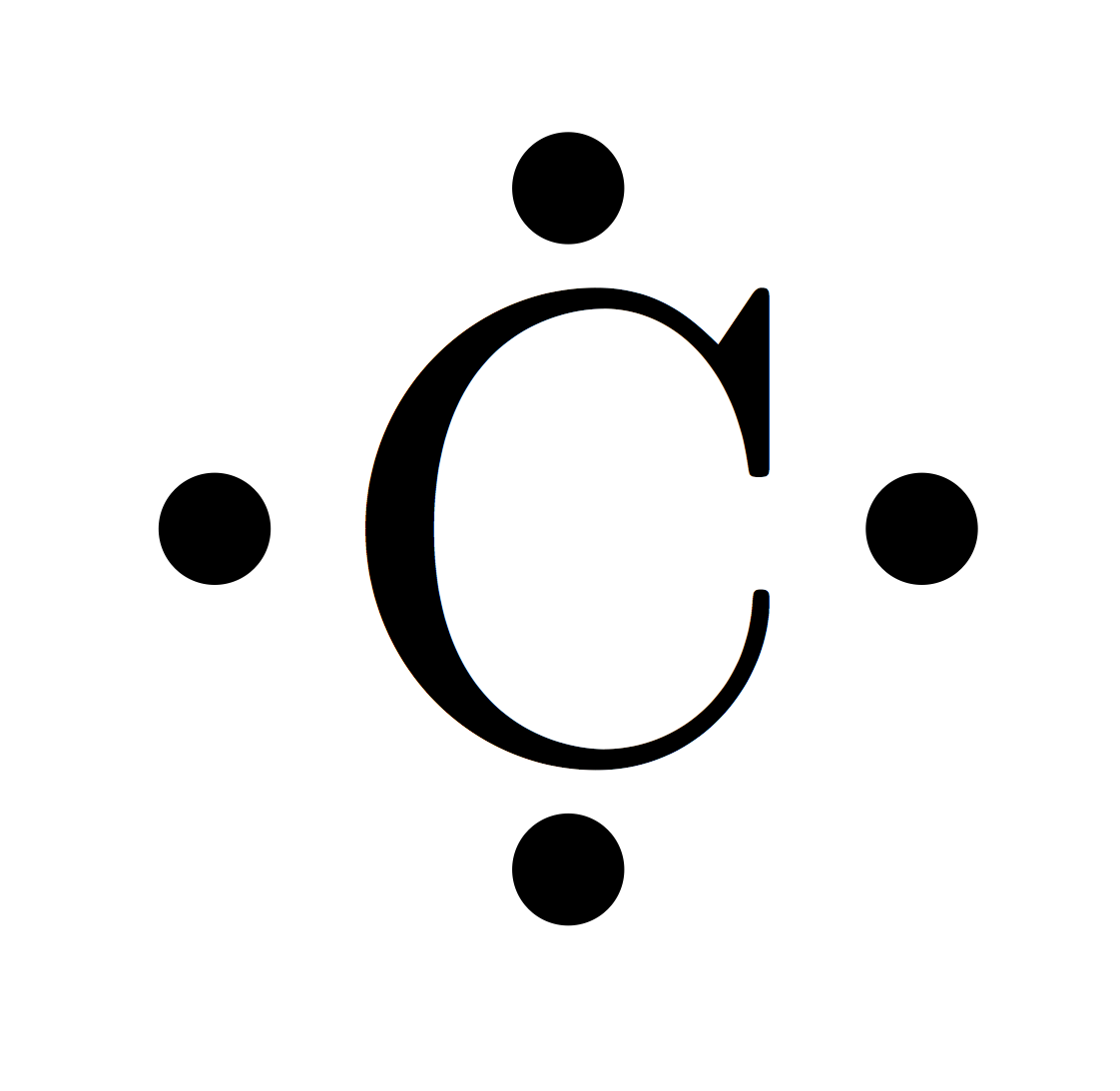
How to Resolve The Valency of Carbon Electronic Configuration
A Carbon atom is a neutral atom that has 6 atomic numbers which imply it has a total of 6 electrons. As per the Aufbau rule, the electrons will be filled into 1s orbital first then 2s, then 2p…so on. Now, for the electron configuration of Carbon, the first 2 electrons will go in 1s orbital since s subshell can hold a maximum of 2 electrons.

Download Carbon Atom Atoms RoyaltyFree Stock Illustration Image Pixabay
Carbon is a chemical element with atomic number 6 which means there are 6 protons in its nucleus. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10-19 coulombs.

Electronic Configuration for Carbon spdf Trick Chemistry Atomic Number 6 YouTube
A step-by-step description of how to write the electron configuration for Carbon (C). In order to write the C electron configuration we first need to know t.
:max_bytes(150000):strip_icc()/carbonatom-58b602855f9b5860464c8bf6.jpg)
Atoms Diagrams Electron Configurations of Elements
Figure \(\PageIndex{2}\): Orbital configuration for carbon atom. (Credit: Joy Sheng; Source: CK-12 Foundation; License: CC BY-NC 3.0(opens in new window)) According to the description of valence bond theory so far, carbon would be expected to form only two bonds, corresponding to its two unpaired electrons.

Orbital Diagram For Carbon (C) Carbon Electron Configuration
The electron configuration of an atom is the representation of the arrangement of electrons distributed among the orbital shells and subshells. Commonly, the electron configuration is used to describe the orbitals of an atom in its ground state, but it can also be used to represent an atom that has ionized into a cation or anion by compensating.
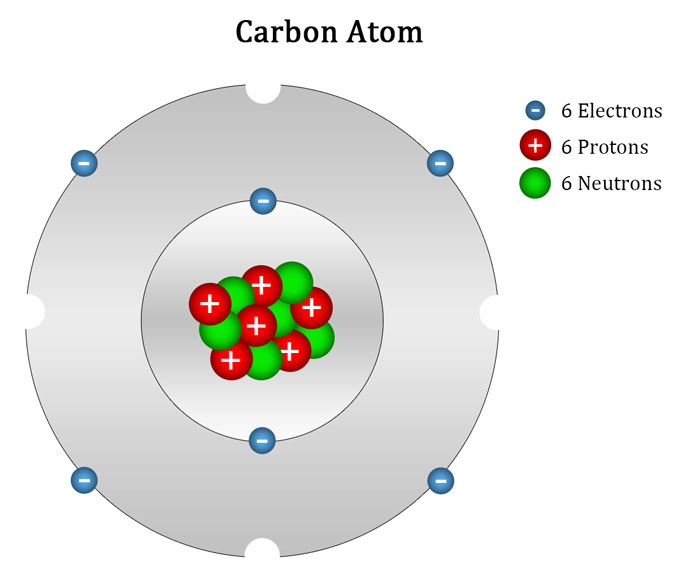
Carbon Atom Ascension Glossary
The electron configuration of carbon is [ He] 2s 2 2p 2 , if the electron arrangement is through orbitals. Electron configuration can be done in two ways. Electron configuration through orbit (Bohr principle) Electron configuration through orbital (Aufbau principle) Carbon atom electron configuration
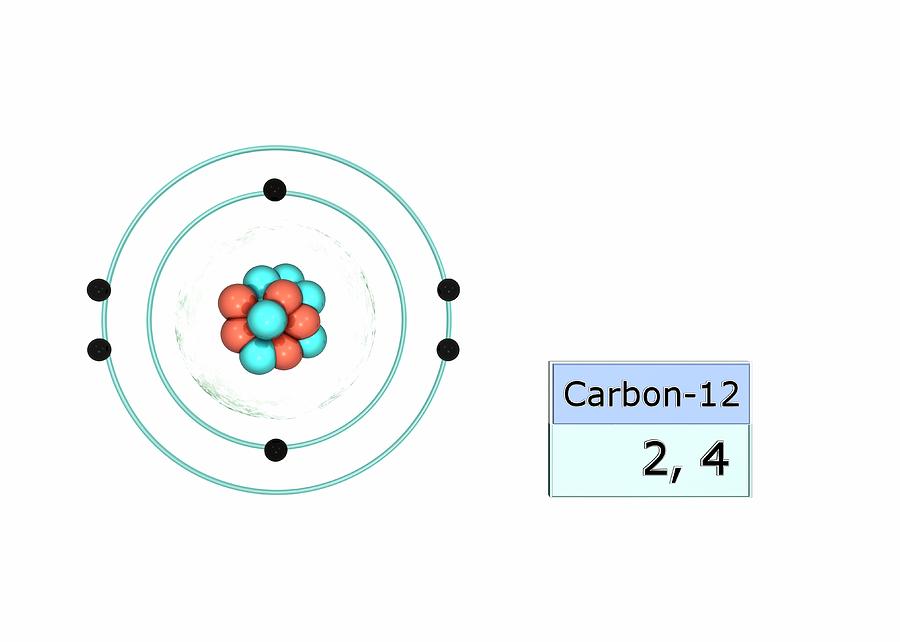
Carbon Electron Configuration Photograph by Photo Libary Pixels
About Transcript Electron configurations describe where electrons are located around the nucleus of an atom. For example, the electron configuration of lithium, 1s²2s¹, tells us that lithium has two electrons in the 1s subshell and one electron in the 2s subshell. Created by Sal Khan. Questions Tips & Thanks Want to join the conversation? Sort by:
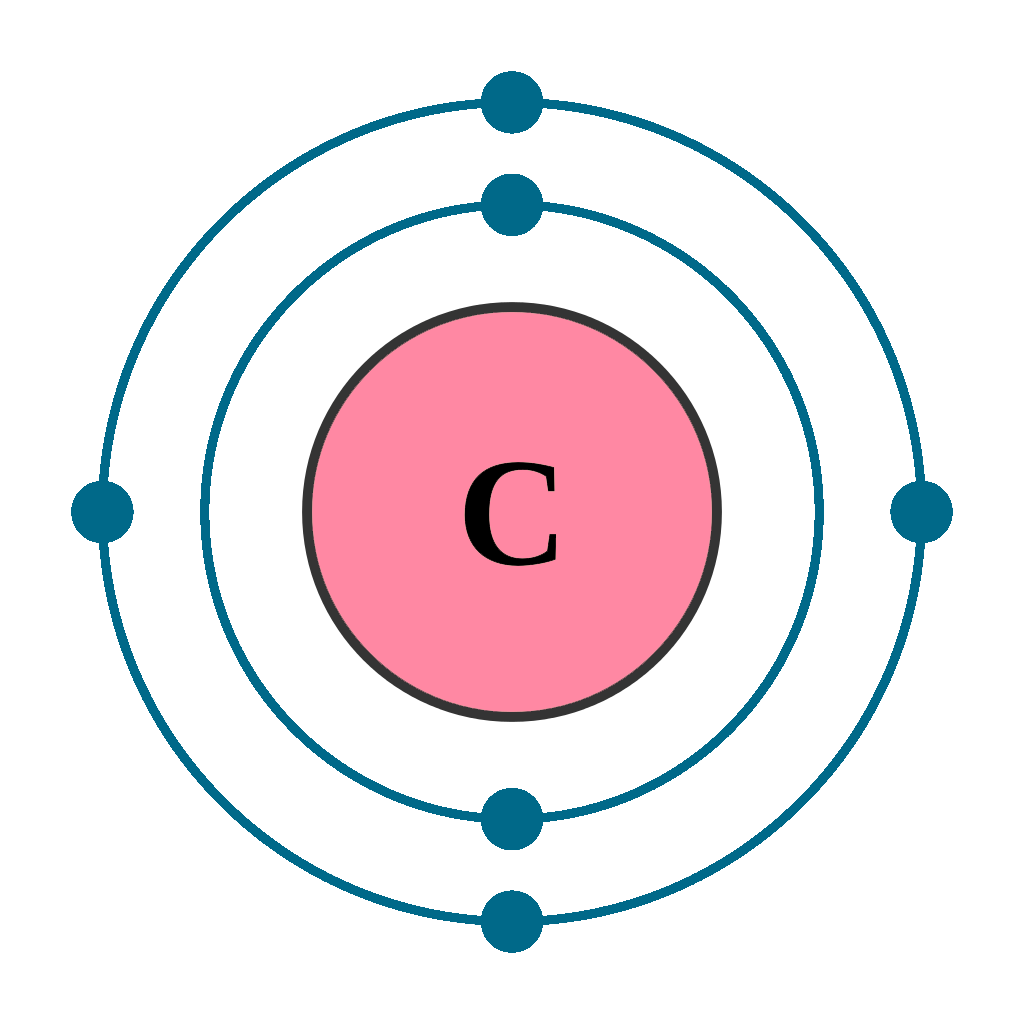
Carbon Element With Reaction, Properties, Uses, & Price Periodic Table
For hydrogen, therefore, the single electron is placed in the 1 s orbital, which is the orbital lowest in energy (Figure 6.29"), and the electron configuration is written as 1 s1 and read as "one-s-one.". A neutral helium atom, with an atomic number of 2 ( Z = 2), has two electrons. We place one electron in the orbital that is lowest in.
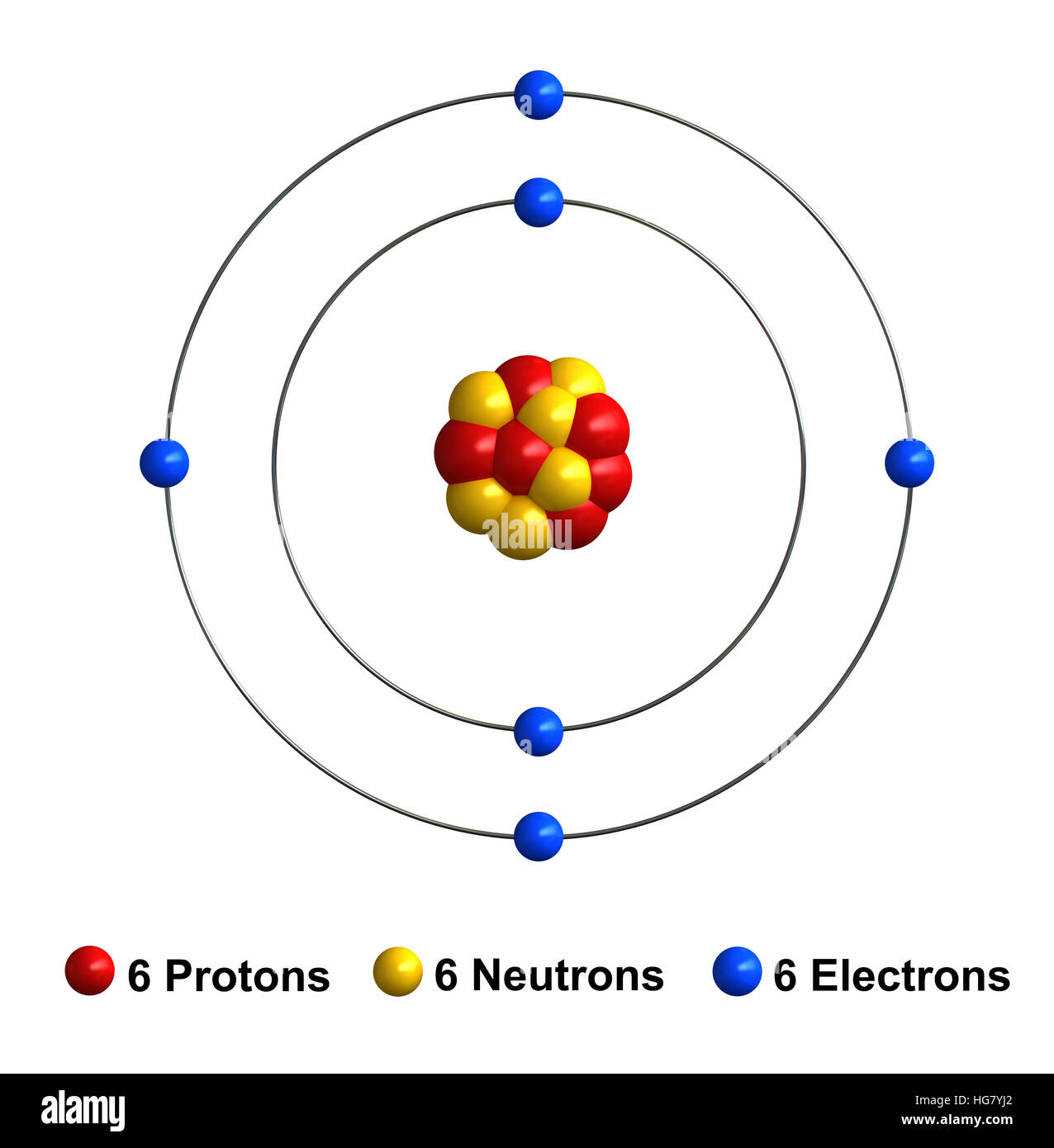
3d render of atom structure of carbon isolated over white background Stock Photo Alamy
Carbon Energy Levels after Rohlf. The ground state electron configuration of carbon is 1s 2 2s 2 2p 2.For excited states, the most typical situation is that five of the electrons maintain the configuration 1s 2 2s 2 2p 1 and a single electron is elevated. The states in the above diagram use the spectroscopic notation to characterize the state of that one electron.
Electron Configuration Electron Shell Valence Electron Carbon, PNG, 800x800px, Electron
Electronic configuration of the Carbon atom. Valence electrons. Orbital diagram. Carbon electron configuration. ← Electronic configurations of elements . C (Carbon) is an element with position number 6 in the periodic table. Located in the II period. Melting point: 3550 ℃.

Carbon12 Electron configuration, Carbon element, Atom
The arrangement of electrons in the orbitals of an atom is called the electron configuration of the atom. We describe an electron configuration with a symbol that contains three pieces of information ( Figure 6.25 ): The number of the principal quantum shell, n,